
Essential Formulae:
E p = mass x gravity x height
E k = 0.5 m v2
E e = Pt = (IV) x t
P = IV
V = IR
P = I 2 R
P = V 2 / R
R = (R1R2) / (R1 + R2) for 2 resistors in parallel
E th = mc (T2 - T1)
and generally E (J) = mc (T2 - T1) = Pt
Quick conversions
1 kWh = 3 600 000 J = 3.6 MJ
1 m3 = 1000 lt = 1000 kg mass
c H2O = 4200 J kg -1 oC -1
1 gallon of petrol or oil = 50 kWh = 180 MJ energy
1 tonne coal = 8000 kWh = 29000 MJ
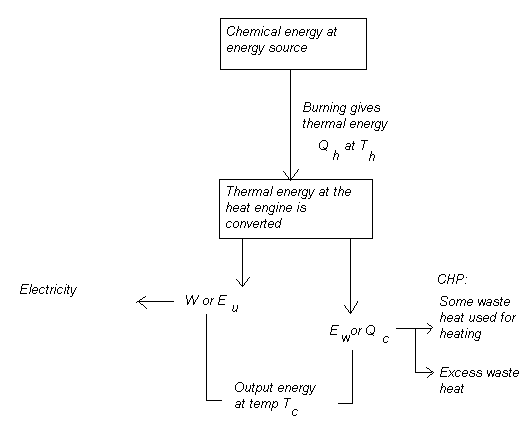
E in = E u + E w
where E in is total energy input to the system, E u is useful energy output and E w is wasted excess (heat) energy.
The 2nd Thermodynamics Law and Equations for engine efficiency:
Efficiency = Useful energy output / Total energy input x 100 = %
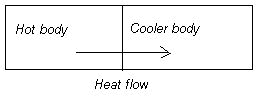
The 2nd Law of Thermodynamics states that heat flows from hotter bodies to cooler bodies:
And only a portion of total input thermal energy can be utilised as useful output kinetic energy as work, while the rest is excess heat energy bi - product of the energy conversion process:
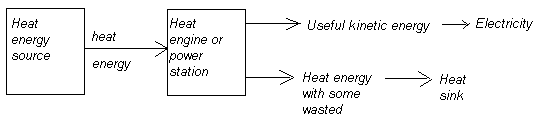
Engine efficiency depends on the temperature at the heat energy source T h and at the heat sink T c, or surrounding system environment. The formula for expressing efficiency is:
where T c can never be 0 K in reality. Note that all formulae should be calculated in Kelvin.
Miscellaneous formulae:
1 N = 1 kg x 1 m s -2 = 1 kg m s -2
F = ma, and for freefalling bodies, F = mg
Work = Force x Distance moved by force
1 J = 1 N x 1 m = 1 kg m s -2 x 1 m
Efficiency = Useful energy output (work) / Total energy input (heat)
or W / Q h, and Q h = Q c + W
For a conventional power station:
At the boiler: n = Thermal energy of steam / Chemical energy of the fuel
At the steam turbine: n = Kinetic energy transferred at the rotor / Thermal energy of the incoming steam
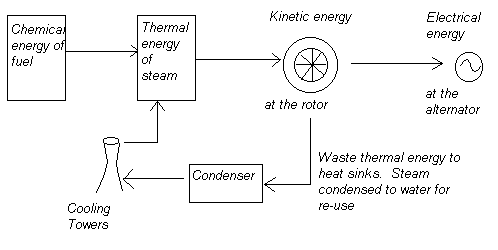
In reality there are several stages of temperature and pressure variation for the steam during the process, and generally the higher the pressure, the hotter the steam and the more kinetic energy available. However, temperature, pressure and kinetic energy become disapated at the turbine.
Project appraisal
Final amount = initial amount x (1 + R) n for compound interest where R is the a.p.r. over n years
Final amount = initial amount x (1 + R) -n for discounted cash flow or depreciation
So Efficiency = (Qh - Q c) / Q h = 1 - (Q c / Q h)
For an ideal heat engine (Carnot engine):
n max = 1 - (T c / T h)
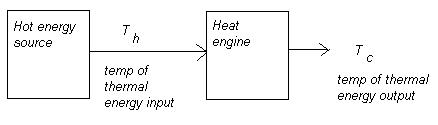
For real engines n > 1 - (T c) / T h) where T c can never be 0 K as stated earlier.
The 2nd Law of Thermodynamics is also the reason one cannot travel backward in time. As heat in a closed system increases, so too does entropy which also increases with time. Entropy in a closed finite system such as the universe (see Laws of Conservation for mass and energy among others) cannot be reversed by 'winding back' time. This is the Thermodynamic Time Arrow. For example, it would be virtually impossible to get back all of the original chemical energy from the burned fuel used to power a heat engine. There are current theories which hypothesize that in imaginary time it might be theoretically possible to go backward, because movement in imaginary time is synonymous with going backward in space-time (see also complex numbers).